|
Infact alkalis are soluble bases, and they are proton acceptors in solutions. For more information, visit Creative Commons Attribution 3.0 Unported. HCl H+ & Cl- On the other hand, bases and alkalis are opposite to acids. V(NaOH) volume of NaOH (aq) in the calorimeter 50.0 mL V(HCl) volume of HCl (aq) added to achieve neutralisation 50.0 mL c(NaOH) concentration of NaOH (aq) 1.00 mol L -1 c(HCl) concentration of HCl (aq) 1. These unbranded versions of the same content are available for you to share, adapt, transform, modify or build upon in any way, with the only requirement being to give appropriate credit to Siyavula. For more information, visit Creative Commons Attribution-NoDerivs 3.0 Unported.įind out more here about the sponsorships and partnerships with others that made the production of each of the open textbooks possible. The only restriction is that you cannot adapt or change these versions of the textbooks, their content or covers in any way as they contain the relevant Siyavula brands, the sponsorship logos and are endorsed by the Department of Basic Education. You can burn them to CD, email them around or upload them to your website. What is a Titration A titration is an analytical procedure used to determine the concentration of a sample. 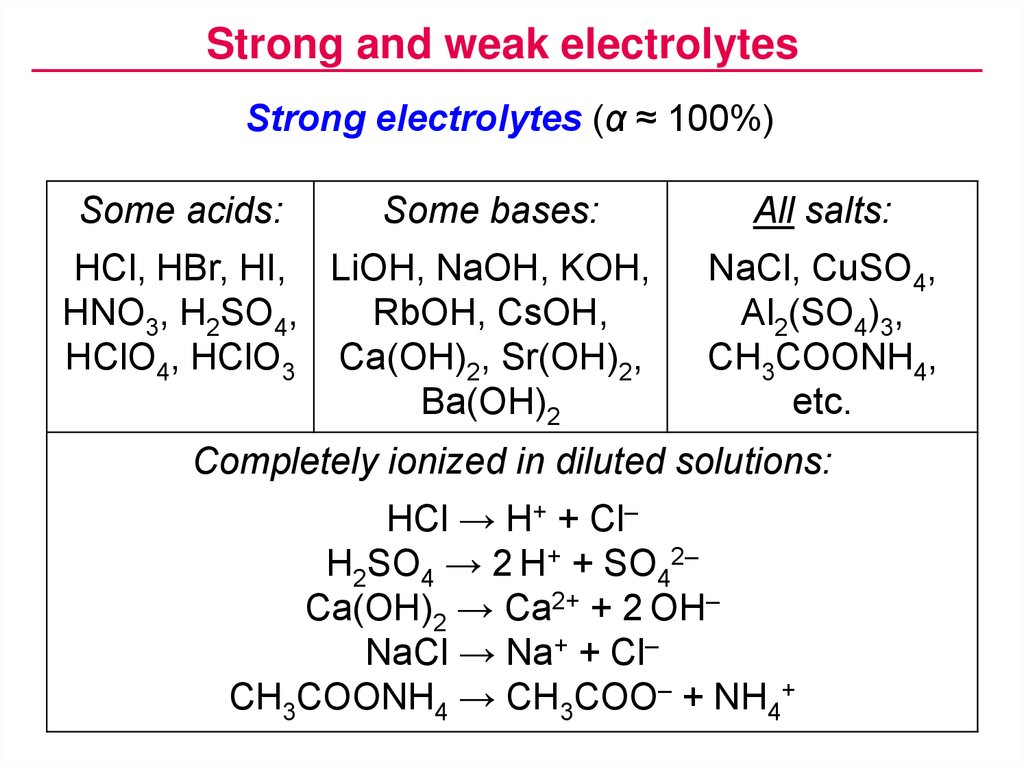
You can download them onto your mobile phone, iPad, PC or flash drive. Using Acid/Base Chemistry: A Quality Control Test. You can photocopy, print and distribute them as often as you like. Sodium hydroxide ionizes in water to form sodium ions and hydroxide ions sulfuric acid ionizes to form hydrogen ions and sulfate ions. The progress of an acid-base titration is often monitored by. You are allowed and encouraged to freely copy these versions. concentration) can be calculated at the equivalence point. The following equation will help you to know how the base gets dissociated in an aqueous solution. 
The compounds with a pH value above 7 are known as bases. Like acids, bases can also be measured by their pH value. 
Better than just free, these books are also openly-licensed! The same content, but different versions (branded or not) have different licenses, as explained: CC-BY-ND (branded versions) Base Bases when dissolved in water will dissociate and release OH- ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
